Engineering's greatest challenge: Our survival
Panel puts 14 jobs on to-do list, ranging from clean water to clean fusion
By Alan Boyle
Science editor
updated 5:08 p.m. ET, Fri., Feb. 15, 2008
BOSTON - After a year of deliberation, an all-star team of technologists on Friday laid out their list of the 21st century's top engineering challenges ?- a list that lifts engineers out of their geeky stereotypes and puts them at the forefront of change.
The Grand Challenges for Engineering call for countering global warming, harnessing nuclear fusion, heading off terrorism, rebuilding cities and reverse-engineering the brain. And those are just a few of the 14 items on the to-do list.
"Meeting some of these is simply imperative for our survival on this planet," Charles Vest, president of the National Academy of Engineering, told reporters. "Some will make us more secure against both human and natural threats. And all will improve the quality of life in our nation and the world."
At the request of the National Science Foundation, the academy brought together a committee of experts to sift through suggestions solicited from engineers, scientists, policymakers and the general public. Among the experts were robotics whiz Dean Kamen, futurist/inventor Ray Kurzweil, Google co-founder Larry Page and genomics pioneer J. Craig Venter.
"Tremendous advances in quality of life have come from improved technology in areas such as farming and manufacturing," Page said in an academy statement listing the challenges. "If we focus our effort on the important grand challenges of our age, we can hugely improve the future."
The committee was chaired by former Defense Secretary William Perry, who is now an engineering professor at Stanford University. Perry noted that the academy's list of 21st-century challenges follows up on an earlier list of the past century's greatest achievements.
"All of these greatest achievements of the 20th century did have a dark side to them," Perry said. For example, the automobile and the airplane sped up the pace of global transport, but also contributed to global warming. Computers opened the way for today's information society, but also opened the way for viruses and cyber-scams.
"We should not only look at how we seize the opportunities of new technology, but how we meet the challenges that were created by the developments in the 20th century," Perry said.
The challenges fall into four general themes: promoting sustainable technologies, advancing human health, reducing vulnerability to threats and increasing the joy of living. The experts decided against focusing on detailed predictions or gadgetry, but instead identified 14 objectives that would benefit humanity and the planet.
The 14 Grand Challenges for Engineering, announced in Boston at the annual meeting of the American Association for the Advancement of Science, are:
Making solar energy affordable: How do you convert and store the power of sunshine at a cost competitive with fossil fuels?
Providing energy from fusion: How do you sustain a controlled fusion reaction for commercial power generation?
Developing carbon sequestration methods: How do you capture the carbon dioxide produced from fossil-fuel burning, and confine that excess carbon underground?
Managing the nitrogen cycle: How do you develop countermeasures for fertilizer use, internal combustion and other activities that contribute to pollution?
Providing access to clean water: How do you address the short supply of water for personal use and irrigation in many areas of the world?
Restoring and improving urban infrastructure: How do you renew aging infrastructure while bringing cities into better ecological balance?
Advancing health informatics: How do you identify the specific factors behind wellness and illness, and follow through on the promise of personalized medicine?
Engineering better medicines: How do you find new treatments for age-old scourges as well as newly emerging diseases?
Reverse-engineering the brain: How do you unlock the secrets of brain function, to heal human diseases and advance the field of artificial intelligence?
Preventing nuclear terror: How do you head off threats from agents who are bent upon bringing ruin to industrial society?
Securing cyberspace: How do you protect the global information infrastructure from identity theft, viruses and other threats without bogging down the flow of data?
Enhancing virtual reality: How do you use computer technology to create imaginative environments for education and entertainment?
Advancing personalized learning: How do you move from a "one-size-fits-all" style of education to more engaging, computer-enhanced teaching techniques?
Engineering the tools for scientific discovery: How do you improve our methods for exploring the frontiers of life, the atom and the cosmos?
The committee decided not to rank the challenges. Instead, the academy is asking the public to vote for the most important challenge and provide comments at the project Web site. The site also offers an overview of the project, excerpts from interviews with committee members and other background information.
Kurzweil, who has already predicted that humanity was heading toward an information-driven "singularity" in the year 2029, said he was excited about the challenges.
"Only technology has the scale to address the pressing problems we have," he said.
Kurzweil focused on the promise of solar power, saying that he expected the cost per watt of sun-generated electricity to equal the cost of fossil-fuel energy in five years. In his view, that would set up a "tipping point," potentially leading to an all-solar energy economy in 20 years. Similar advances could be anticipated in neuroscience, genetics and medicine as those fields moved closer to the model set by information technology, he said.
Calestous Juma, a Harvard professor who focuses on international development, said the challenges reflected a "more enlightened understanding" of the role played by technology in an increasingly globalized society. He hoped the project would benefit developing regions as well as the world's more industrialized countries. Some of the challenges target problems that have long bedeviled developing countries, such as water shortages and the scourge of malaria.
Vest told msnbc.com that the academy would use the challenges internally "as organizing principles for our own programs," and he hoped the project would draw the attention of candidates from both parties in this election year.
"In the 21st century, you're not going to be able to lead effectively unless the government at the highest level draws on the power of science and engineering and medicine," he said.
Perry said he supported the idea of holding a presidential debate after the nominating conventions that would focus on scientific and engineering challenges. "The next administration has a lot to do with this, and we will be trying to influence their decisions early," he said.
Vest also hoped the challenges would provide a wider perspective for the next generation of engineers. In the past, engineering has been looked upon as separate from scientific and medical pursuits, but Vest said the next century would be different. "These areas are merging," he told msnbc.com.
"Engineering is really a way of improving the human condition," Vest said. "I hope that this will inspire a lot of men and women to participate in meeting these challenges."
In addition to Perry, Jura, Kamen, Kurzweil, Page and Venter, members of the academy's committee included:
Alec Broers, chairman of the Science and Technology Select Committee in Britain's House of Lords.
Farouk El-Baz, director of Boston University's Center for Remote Sensing.
Wesley Harris, professor of aeronautics and astronautics at the Massachusetts Institute of Technology.
Bernadine Healy, former director of the National Institutes of Health, and currently health editor and columnist at U.S. News & World Report.
W. Daniel Hillis, chairman and co-founder of Applied Minds Inc.
Robert Langer, MIT professor.
Jaime Lerner, architect and urban planner.
Bindu Lohani, director general and chief compliance officer, Asian Development Bank.
Jane Lubchenco, professor of marine biology and zoology at Oregon State University.
Mario Molína, biochemist at the University of California at San Diego.
Robert Socolow, professor of mechanical and aerospace engineering at the Princeton University Environmental Institute.
Jackie Ying, executive director for the Institute of Bioengineering and Nanotechnology.
© 2008 MSNBC Interactive
URL: http://www.msnbc.msn.com/id/23175788/
As to who is right of who in Germany, I have no clue.
miniTAX wrote:The 4th IPCC report builds on the work of earlier reports from which it is concluded that climate change is real and that it stems from human activity.
The IPCC mandate is, I cite:
"to assess on a comprehensive, objective, open and transparent basis the latest scientific, technical and socio-economic literature produced worldwide relevant to the understanding of the risk of human-induced climate change, its observed and projected impacts and options for adaptation and mitigation".
That is akin to studying how many martians landed at Roswell, was it a hundred, or one or two? It would really be a non-starter to have to conclude that none landed there wouldn't it, after all that money spent on such a study, and all the experts receiving funding to study how many landed there? No expert wants to come to a conclusion that essentially says it was a waste of money to pay him to study it.
By the way HB, you never did answer my question. With Arctic ice returning with a vengeance this winter, did your lake freeze over this winter?
Yet a few posts back you were saying even the IPCC cant quantify the magnitude of CO2 change and whether it was human induced or not.
Nobody denies that there is anthropogenic climate change.
I have already shown you wrong.
Lets assume a solution X has 20 grams of a substance Y in it.
Now there is a process that acts on X and removes 19 grams of Y every hour but also returns 19 grams of Y every hour. We will call this process A.
If we monitor X and process B for 5 hours there is no increase of Y in X.
Now we introduce another process B which adds 5 grams of Y every hour and removes none of Y.
If after 5 hours we solution X has 22 grams of substance Y in it where did the extra Y come from? Process A or Process B?
If we know that Process A introduces 5 grams of Y every hour but haven't measured B during that 5 hours what is the logical assumption to make?
We know for a fact that humans introduce MORE CO2 into the atmosphere than the increase we observe. Where does that extra CO2 go? If the oceans and vegetation don't take up MORE CO2 than they put out where is that CO2?
Here is another explanation.
http://www.realclimate.org/index.php?p=87
and another mathematical proof here
http://fermiparadox.wordpress.com/2007/04/21/a-simple-calculation/
Humans do not put out 1/20 of the increase in CO2. They put out 2 or more times the increase in CO2.
http://www.skepticalscience.com/human-co2-smaller-than-natural-emissions.htm
Humans account for 32 gigatonnes per year with the 15 gigatonnes increase per year in the atmosphere.
http://www.gcrio.org/ipcc/qa/05.html
4 lines of evidence show that humans cause CO2 increase.

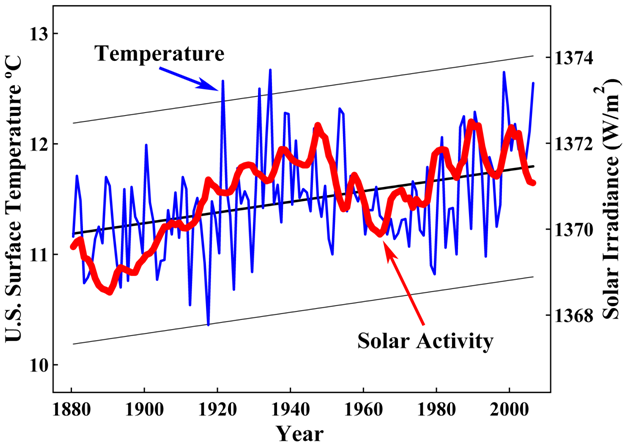
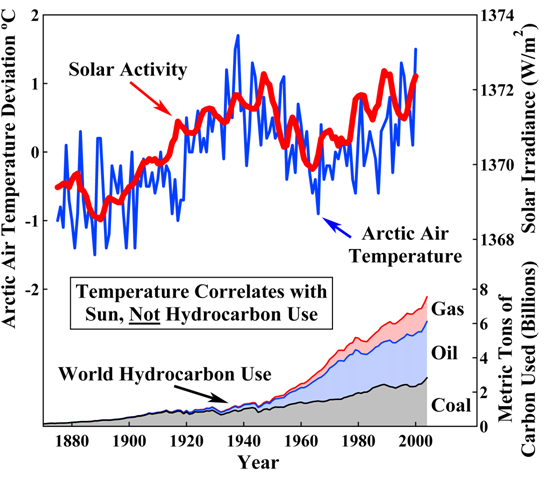
[No! I have already shown you are wrong.
Let's assume mixture A has 100 grams of C in it.
Now there is a process that adds 50 grams of C and removes 50 grams of C from mixture A every hour. We will call this process E-P.
If we monitor A and process E-P for 5 hours we learn that there is no increase or decrease of C in mixture A.
Now we introduce another process H which adds 4 grams of C to mixture A and removes 2 grams from mixture A every hour.
After 5 hours we observe that mixture A has 110 grams of C in it.
Now we introduce another process S which adds 12 grams of C to mixture A and removes 4 grams from mixture A every hour.
After 5 hours we observe that mixture A has 150 grams of C in it.
Obviously S has introduced 40 more grams of C into mixture A.
Let:
A = the atmosphere;
C = CO2;
E-P = the process of evaporation of CO2 out of its surface sink into A and the precipitation of CO2 out of A into its surface sink.
H-E-P = the human process of addiing CO2 into A and the E-P process of removal of 50% of these additions.
S = the process caused by the increase in solar irradiance that increases the temperature of both A and the CO2 surface sink.
CO2 in the atmosphere:
1975 = 330ppm
2005 = 385ppm
FOR THE PERIOD 1975 TO 2005:
%increase in CO2 in the atmosphere = 100% x (385-330)/330 = 16.7%
%increase in average global temperature not caused by solar irradiance increase = 0.1488%.
Ratio of CO2 increase to the average global temperature increase that is not caused by solar irradiance increase = 16.67% / 0.1488% = 112
Looks like average global temperature is relatively insensitive to CO2 content of the atmosphere.
parados wrote:We know for a fact that humans introduce MORE CO2 into the atmosphere than the increase we observe. Where does that extra CO2 go? If the oceans and vegetation don't take up MORE CO2 than they put out where is that CO2?
Parados, not because you keep repeating this same piece of junk science would make it better.
ican711nm wrote:[No! I have already shown you are wrong.
Let's assume mixture A has 100 grams of C in it.
Now there is a process that adds 50 grams of C and removes 50 grams of C from mixture A every hour. We will call this process E-P.
If we monitor A and process E-P for 5 hours we learn that there is no increase or decrease of C in mixture A.
Now we introduce another process H which adds 4 grams of C to mixture A and removes 2 grams from mixture A every hour.
After 5 hours we observe that mixture A has 110 grams of C in it.
Now we introduce another process S which adds 12 grams of C to mixture A and removes 4 grams from mixture A every hour.
After 5 hours we observe that mixture A has 150 grams of C in it.
Obviously S has introduced 40 more grams of C into mixture A.
Let:
A = the atmosphere;
C = CO2;
E-P = the process of evaporation of CO2 out of its surface sink into A and the precipitation of CO2 out of A into its surface sink.
H-E-P = the human process of addiing CO2 into A and the E-P process of removal of 50% of these additions.
S = the process caused by the increase in solar irradiance that increases the temperature of both A and the CO2 surface sink.
your analogy makes no sense.
1. You have no evidence of humans removing CO2 from the atmosphere in quantities similar to your analogy. In fact there is no evidence of humans removing CO2 in any quantity that would be considered substantial.
2. The solar radiation is not a NEW process so can't suddenly be introduced after humans have started pumping CO2 into the air. Solar variation has occurred for billions of years.
3. Solar radiation doesn't create CO2 in the atmosphere so your attempt to use it is completely bogus.
We know that humans pump out more CO2 than the yearly increase. You have provided no other viable source for the increase in CO2. You have provided no viable place for the human CO2 to go so it is NOT the cause of the increase. You have provided no refutation of the KIND of carbon atoms that make up the inrease. Carbon atoms that are most likely to have come from fossil fuels in the quantities observed. You have provided no evidence of any change other than humans for the increase in CO2.
http://co2.cms.udel.edu/Increasing_Atmospheric_CO2.htm
About half of the recent emissions are not accumulating in the atmosphere, but are going into the ocean and, to a lesser extent, into soils. These are considered "sinks" in the global carbon budget because they take up atmospheric CO2.
ican711nm wrote:CO2 in the atmosphere:
1975 = 330ppm
2005 = 385ppm
FOR THE PERIOD 1975 TO 2005:
%increase in CO2 in the atmosphere = 100% x (385-330)/330 = 16.7%
%increase in average global temperature not caused by solar irradiance increase = 0.1488%.
Ratio of CO2 increase to the average global temperature increase that is not caused by solar irradiance increase = 16.67% / 0.1488% = 112
Looks like average global temperature is relatively insensitive to CO2 content of the atmosphere.
No one ever said it was the total reason for the earth's temperature or that increasing it by 50% would increase the earth's temperature by 50% or even 10%.
parados wrote:ican711nm wrote:[No! I have already shown you are wrong.
Let's assume mixture A has 100 grams of C in it.
Now there is a process that adds 50 grams of C and removes 50 grams of C from mixture A every hour. We will call this process E-P.
If we monitor A and process E-P for 5 hours we learn that there is no increase or decrease of C in mixture A.
Now we introduce another process H which adds 4 grams of C to mixture A and removes 2 grams from mixture A every hour.
After 5 hours we observe that mixture A has 110 grams of C in it.
Now we introduce another process S which adds 12 grams of C to mixture A and removes 4 grams from mixture A every hour.
After 5 hours we observe that mixture A has 150 grams of C in it.
Obviously S has introduced 40 more grams of C into mixture A.
Let:
A = the atmosphere;
C = CO2;
E-P = the process of evaporation of CO2 out of its surface sink into A and the precipitation of CO2 out of A into its surface sink.
H-E-P = the human process of addiing CO2 into A and the E-P process of removal of 50% of these additions.
S = the process caused by the increase in solar irradiance that increases the temperature of both A and the CO2 surface sink.
your analogy makes no sense.
1. You have no evidence of humans removing CO2 from the atmosphere in quantities similar to your analogy. In fact there is no evidence of humans removing CO2 in any quantity that would be considered substantial.
2. The solar radiation is not a NEW process so can't suddenly be introduced after humans have started pumping CO2 into the air. Solar variation has occurred for billions of years.
3. Solar radiation doesn't create CO2 in the atmosphere so your attempt to use it is completely bogus.
We know that humans pump out more CO2 than the yearly increase. You have provided no other viable source for the increase in CO2. You have provided no viable place for the human CO2 to go so it is NOT the cause of the increase. You have provided no refutation of the KIND of carbon atoms that make up the inrease. Carbon atoms that are most likely to have come from fossil fuels in the quantities observed. You have provided no evidence of any change other than humans for the increase in CO2.
YOUR INTERPRETATION OF MY POST MAKES ZERO SENSE.
1. I did not claim humans removed CO2 from the atmosphere.
Quote:
http://co2.cms.udel.edu/Increasing_Atmospheric_CO2.htm
About half of the recent emissions are not accumulating in the atmosphere, but are going into the ocean and, to a lesser extent, into soils. These are considered "sinks" in the global carbon budget because they take up atmospheric CO2.
2. The solar irradiation process is a changing process. Sometimes it's level of irradiation is relatively low; sometimes it is average; sometimes it is relatively high. During 1975-2005 it increased to relatively high.
3. Solar radiation heats the surface of the globe. When solar radiation intensity increases, it heats up the surface of the globe more. Our globe's surface, especially the oceans, are CO2 sinks. When they are heated more, they emit more CO2 into the atmosphere. When the atmosphere is warmer, it can retain more CO2 than when it is cooler. Therefore, when solar irradiation increases, it causes more CO2 to be emitted into the atmosphere from surface CO2 sinks, and it causes more CO2 to be retained in the atmosphere.
I have identified an additional source of CO2 in the atmosphere besides humans. That additional source is the natural land and ocean surfaces of the earth.
By the way, most of the CO2 in the oceans arrived their over thousands of years before 1975, even before 1901, even before 1850, even before George Washington allegedly chopped down a cherry tree when a youngster. Wise up! ... if you can ...
...
So where does the human released CO2 go? We know that humans release more than twice the amount of CO2 that is newly retained in the atmosphere. Where does it go? You have never answered that question which is required if you want to claim the source is other than human. I have posted repeated scientific references that show the ocean absorbs more than it releases.
http://co2.cms.udel.edu/Increasing_Atmospheric_CO2.htm
About half of the recent emissions are not accumulating in the atmosphere, but are going into the ocean and, to a lesser extent, into soils. These are considered "sinks" in the global carbon budget because they take up atmospheric CO2.
