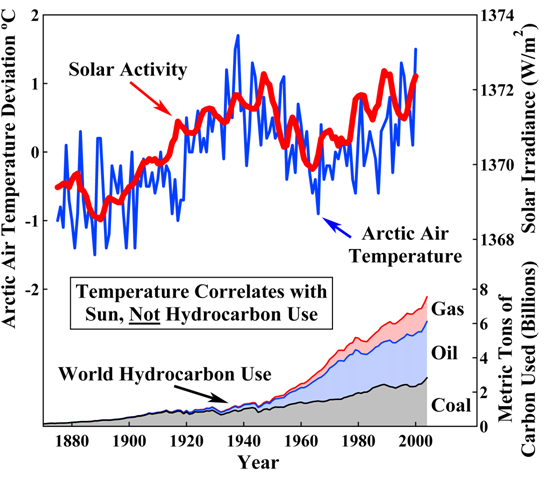
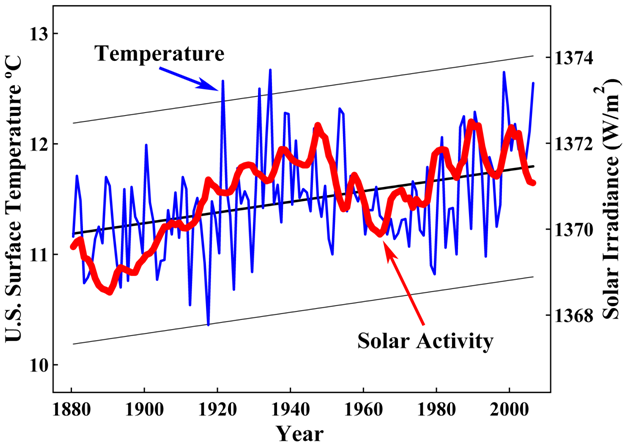
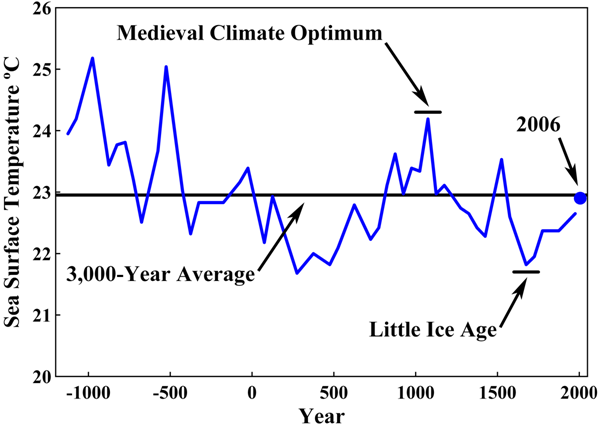
minitax, you asked for ONE measurement. How about 72,000? Your claim that acidification hasn't been measured is, yes, all wet.
source:
http://www.noaanews.noaa.gov/stories2004/s2261.htm
STUDY DETAILS DISTRIBUTION, IMPACTS OF CARBON DIOXIDE IN THE WORLD OCEANS
July 15, 2004 ?- An international team of scientists has completed the first comprehensive study of the ocean storage of carbon dioxide derived from human activity based on a decade-long survey of global ocean carbon distributions in the 1990s. The global survey combined measurements of carbon and other ocean factors such as temperature, salinity, oxygen, nutrients and chlorofluorocarbon tracers in the Atlantic, Pacific and Indian oceans to determine that the oceans have taken up about 118 billion metric tons of carbon dioxide from human sources (anthropogenic CO2) between 1800 and 1994. This is approximately a third of its long-term potential. (Click NOAA image for larger view of map showing total inventory of human-caused C02 is found in the North Atlantic in 1994. Click here for high resolution version, which is a large file. Click here for larger version at 3,000 x 1,633 pixels at 300 dpi. Please credit "NOAA.")
These findings, plus a companion paper on the impacts of anthropogenic CO2 on the chemistry of the oceans and the potential response of certain marine species to the changes in CO2 levels, will be published in the July 16 issue of the journal Science. Lead authors for both papers are scientists with NOAA.
Analysis of CO2 levels in ice cores have shown scientists that for the 400,000 years before the industrial revolution began in the 1800s, atmospheric CO2 concentrations remained between 200 and 280 parts per million. Today CO2 levels are reaching 380 parts per million in the atmosphere.
"If the ocean had not removed 118 billion metric tons of anthropogenic carbon between 1800 and 1994, the CO2 level in the atmosphere would be about 55 parts per million greater than currently observed," said Christopher Sabine an oceanographer at the NOAA Pacific Marine Environmental Laboratory in Seattle, Wash., and the lead author of one of the papers.
Because the ocean mixes slowly, the anthropogenic CO2 from the atmosphere is generally confined to the upper layers of the ocean.
"About half of the anthropogenic CO2 taken up over the last 200 years can be found in the upper 10 percent of the ocean. The ocean has removed 48 percent of the CO2 we have released to the atmosphere from burning fossil fuels and cement manufacturing," Sabine said.
Sabine and his colleagues from the United States, South Korea, Australia, Canada, Japan, Spain, and Germany reviewed data gathered during the 1990s as part of three major research programs: the World Ocean Circulation Experiment (WOCE), the Joint Global Ocean Flux Study (JGOFS) and NOAA's Ocean-Atmosphere Carbon Exchange Study (OACES).
This new global data set of ocean-carbon system observations, co-sponsored in the United States by NOAA, National Science Foundation and Department of Energy, is unprecedented with more than 72,000 carbon measurements, 10 times more observations than the previous global survey in the 1970s and 10 times more accurate.
"This research presents the first complete synthesis of modern global ocean inorganic carbon measurements," said James Yoder, director of NSF's ocean sciences division.
There are two large reservoirs of carbon that are capable of taking significant amounts of CO2 out of the atmosphere: the ocean and land plants. Studies over the last decade have indicated that the land plants are taking up CO2 at rates comparable to the oceans. The new high quality ocean carbon measurements allow scientists to determine that over a 200-year time-scale, the land plants have released more CO2 to the atmosphere than they have taken up. Over the long-term, therefore, the ocean has been the only reservoir to consistently take up anthropogenic CO2 from the atmosphere.
The uptake of anthropogenic CO2 by the ocean changes its chemistry and potentially can have significant impacts on the biological systems in the upper oceans.
Richard Feely, a marine chemist with the NOAA Pacific Marine Environmental Laboratory, and colleagues describe two major impacts of the oceanic uptake of anthropogenic CO2. First, they demonstrated that a substantial amount of the calcium carbonate shells produced in surface waters dissolves in the upper ocean. Second, they summarized the available evidence on the response of marine calcifying organisms to elevated CO2.
Feely noted that scientists have seen a reduced ability to produce protective calcium carbonate shells in many species of marine organisms at high CO2 levels, including corals and free-swimming algae (plant-like organisms) and animals on which other marine life feed. Recent studies have shown that calcification rates can drop by as much as 25 percent to 45 percent at CO2 levels equivalent to atmospheric concentrations of 700 to 800 parts per million that will be reached by the end of the century if fossil fuel consumption continues at projected levels.
The scientists note that the dissolving calcium carbonate shells also partially act to neutralize the CO2, thus allowing the ocean to take up more carbon dioxide from the atmosphere. However, the effects of decreased calcification in microscopic algae and animals could alter marine food webs and, combined with other changes in salinity, temperature and upwelled nutrients, could substantially alter the diversity and productivity of the ocean.
NOAA is dedicated to enhancing economic security and national safety through the prediction and research of weather and climate-related events and providing environmental stewardship of the nation?s coastal and marine resources. NOAA is part of the U.S. Department of Commerce.
Relevant Web Sites
NOAA CO2 Program
NOAA Pacific Marine Environmental Laboratory
NOAA Research
Media Contact:
Jana Goldman, NOAA Research, (301) 713-2483 ext. 181